 The overall shape of an organic molecule is fixed by the shape of the central carbon. With fewer 90° LP–BP repulsions, we can predict that the structure with the lone pair of electrons in the equatorial position is more stable than the one with the lone pair in the axial position. Looking at its Lewis structure we can state that molecular geometry of PH3 is trigonal pyramidal. Pcl3 Shape And Bond Angle Why PH3 is non polar While PCl3 is polar. If we place it in the axial position, we have two 90° LP–BP repulsions at 90°. 
The molecular form is trigonal bipyramidal with a bond angle of 175. B.Trigonal bipyramidal geometry is represented by chlorine trifluoride (ClF3). For the PH3 molecule, the bond angles are roughly 93.5. However, because the axial and equatorial positions are not chemically equivalent, where do we place the lone pair? If we place the lone pair in the equatorial position, we have three LP–BP repulsions at 90°. 160 views, 4 likes, 0 loves, 0 comments, 0 shares, Facebook Watch Videos from Geometry Of Molecules: PH3 Molecular Geometry Shape and Bond Angles (Phosphorus Trihydride) PH3 is one of the easy. The center atom has a lone pair of electrons and makes three sigma bonds with hydrogen atoms. There are three P-H bonds at the PH3 molecular geometry. The PH3 molecule has a tetrahedral geometry shape because it contains three hydrogen atoms. 355 Sample Exercises 9.3 and Practice Exercises 1 & 2. B.Trigonal bipyramidal geometry is represented by chlorine trifluoride. The H- P-H bond angle is 107 degrees in the tetrahedral molecular geometry. The hybridization of the oxygen atom labeled y in the structure below is. 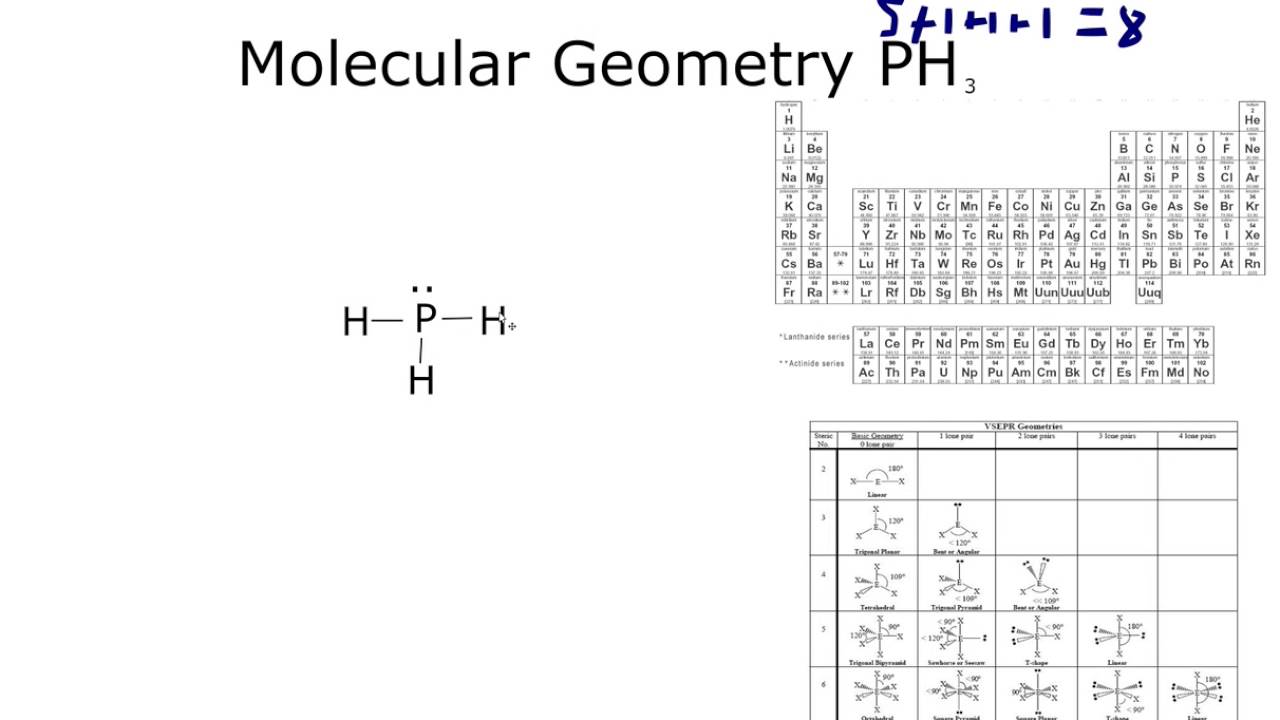
We designate SF 4 as AX 4E it has a total of five electron pairs. For the PH3 molecule, the bond angles are roughly 93.5. 
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
